Swallowing and Nutrition
Most patients with ALS develop swallowing difficulties, which may increase the risk of malnutrition, weight loss, and further illnesses (Radunović et al., 2007). Early management of dysphagia, beginning with a thorough swallowing evaluation that will lead to a treatment plan, is critical to improving quality of life and survival advantage in patients with ALS.
Evaluation
Assessment should begin with a clinical examination of the patient’s swallowing functions using a Fiberoptic Endoscopic Examination of Swallowing (FEES) and Modified Barium Swallow (MBS), and making careful observations when eating solids and drinking liquids.
A) FEES- Even in the absence of patient complaints, dysphagia is a frequent comorbidity. According to D’Ottaviano et al. (2013), while only 72.79% of patients with ALS complained of dysphagia, endoscopic examination revealed that all patients had some form of swallowing alteration, therefore making it “mandatory for dysphagia to be investigated in cases of ALS”.
B) MBS- An MBS can provide an image of the entire duration of the swallow and enable the complete oropharyngeal tract to be viewed. Although, due to respiratory dysfunction and high risk of aspiration in patients with ALS, every precaution should be taken to avoid barium aspiration as this may cause increased patient stress (Kühnlein et al., 2008).
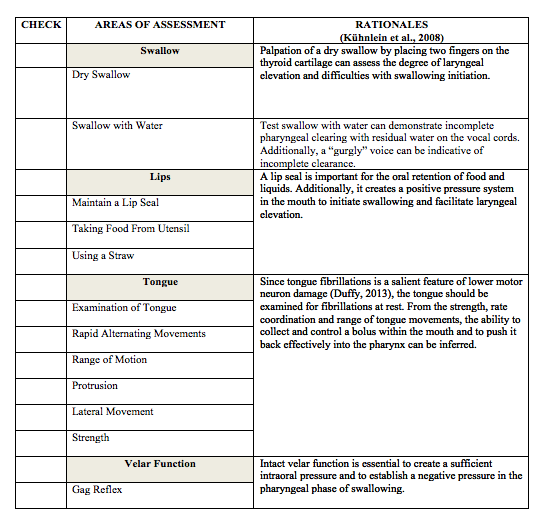
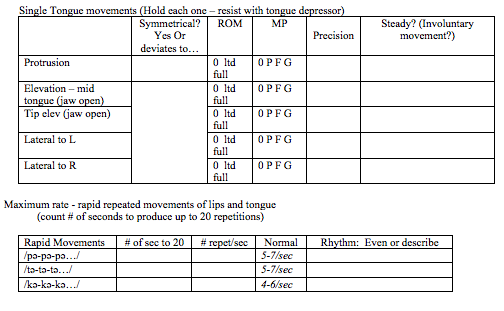
C) Clinical Observations- Kuhnlein et al., (2008) presents several areas of swallowing and oral motor facial movements and structures that should be targeted in a clinical assessment. A table has been created and organized based on Kuhnlein et al., (2008)’s recommended areas of assessment in order to better assist the clinician with data recording (Please see Table 1). Additionally, certain sections from Langmore 2013 Dyarthria Clinical Evaluation have been extracted and modified to assist the clinician in assessing those targeted swallowing areas.
Swallowing Strategies:
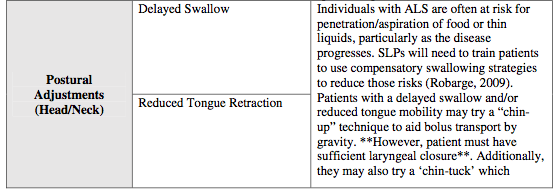
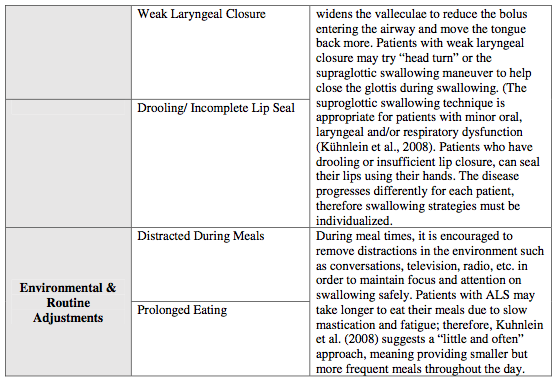
The complementary use of FEES, MBS, and clinical observations can generate a plan for swallowing therapy. Given that ALS is a progressive disease, therapy should focus on developing compensatory strategies such as bolus/diet modifications, body positioning strategies, postural and environmental/routine adjustments, using a multidisciplinary approach to ensure a safer swallow (Robarge, 2009). (Please see Table 2)
Nutrition:
Given the progression of the disease, these swallowing interventions will eventually stop working and dysphagia, weight loss, and malnutrition will predominate. At this stage, alternative feeding methods such as a percutaneous endoscopic gastronomy (PEG) tube may be considered to maintain adequate nutrition. PEG is a supplemental feeding tube that is placed through the abdominal wall to the stomach for a direct food source (Albert, Murphy, Del Bene, Rowland, & Mitsumoto, 2001; Katzberg & Benatar, 2011; Langmore, Kasarskis, Manca, & Olney, 2006).
According to Albert et al. (2001), PEG use is part of a broad and proactive approach to disease management undertaken by patients and their families. A Cochrane Review examined the efficacy of PEG placement on the survival rate measured in months; nutritional status measured by weight change or body mass index; self-perceived quality of life; and complications of PEG in patients with ALS (Katzberg & Benatar, 2011; Langmore et al., 2006). There are currently no randomized clinical trials that compare enteral feeding to those patients who continue with oral feeding, therefore conclusions are tentative. The patient’s QOL is understudied and there is no consensus in the literature for the best timing of PEG insertion. However, the findings do suggest a possible survival advantage and potential support for improved nutrition.
Evaluation
Assessment should begin with a clinical examination of the patient’s swallowing functions using a Fiberoptic Endoscopic Examination of Swallowing (FEES) and Modified Barium Swallow (MBS), and making careful observations when eating solids and drinking liquids.
A) FEES- Even in the absence of patient complaints, dysphagia is a frequent comorbidity. According to D’Ottaviano et al. (2013), while only 72.79% of patients with ALS complained of dysphagia, endoscopic examination revealed that all patients had some form of swallowing alteration, therefore making it “mandatory for dysphagia to be investigated in cases of ALS”.
B) MBS- An MBS can provide an image of the entire duration of the swallow and enable the complete oropharyngeal tract to be viewed. Although, due to respiratory dysfunction and high risk of aspiration in patients with ALS, every precaution should be taken to avoid barium aspiration as this may cause increased patient stress (Kühnlein et al., 2008).
C) Clinical Observations- Kuhnlein et al., (2008) presents several areas of swallowing and oral motor facial movements and structures that should be targeted in a clinical assessment. A table has been created and organized based on Kuhnlein et al., (2008)’s recommended areas of assessment in order to better assist the clinician with data recording (Please see Table 1). Additionally, certain sections from Langmore 2013 Dyarthria Clinical Evaluation have been extracted and modified to assist the clinician in assessing those targeted swallowing areas.
Swallowing Strategies:
The complementary use of FEES, MBS, and clinical observations can generate a plan for swallowing therapy. Given that ALS is a progressive disease, therapy should focus on developing compensatory strategies such as bolus/diet modifications, body positioning strategies, postural and environmental/routine adjustments, using a multidisciplinary approach to ensure a safer swallow (Robarge, 2009). (Please see Table 2)
Nutrition:
Given the progression of the disease, these swallowing interventions will eventually stop working and dysphagia, weight loss, and malnutrition will predominate. At this stage, alternative feeding methods such as a percutaneous endoscopic gastronomy (PEG) tube may be considered to maintain adequate nutrition. PEG is a supplemental feeding tube that is placed through the abdominal wall to the stomach for a direct food source (Albert, Murphy, Del Bene, Rowland, & Mitsumoto, 2001; Katzberg & Benatar, 2011; Langmore, Kasarskis, Manca, & Olney, 2006).
According to Albert et al. (2001), PEG use is part of a broad and proactive approach to disease management undertaken by patients and their families. A Cochrane Review examined the efficacy of PEG placement on the survival rate measured in months; nutritional status measured by weight change or body mass index; self-perceived quality of life; and complications of PEG in patients with ALS (Katzberg & Benatar, 2011; Langmore et al., 2006). There are currently no randomized clinical trials that compare enteral feeding to those patients who continue with oral feeding, therefore conclusions are tentative. The patient’s QOL is understudied and there is no consensus in the literature for the best timing of PEG insertion. However, the findings do suggest a possible survival advantage and potential support for improved nutrition.
Table 1:
I. LIPS
MP Scoring Key
Good = can resist opposing pressure
Fair = cannot resist opposing pressure
Poor = cannot move full range
None = none or trace movement
MP Scoring Key
Good = can resist opposing pressure
Fair = cannot resist opposing pressure
Poor = cannot move full range
None = none or trace movement
I. TONGUE
Observe tongue resting in mouth:
Symmetry _______________ size/ bulk/ tone: ______________ steady? __________
Observe tongue resting in mouth:
Symmetry _______________ size/ bulk/ tone: ______________ steady? __________
I. VELOPHARYNGEAL SYSTEM
Observe velopharyngeal area at rest: _______________________________________
Observe velopharyngeal closure:
For “ah” once symmetry / deviate to (L/ R)
For “ah” repeated symmetry / deviate to (L/ R)
Gag reflex: present / absent R/ L (optional task)
Observe velopharyngeal area at rest: _______________________________________
Observe velopharyngeal closure:
For “ah” once symmetry / deviate to (L/ R)
For “ah” repeated symmetry / deviate to (L/ R)
Gag reflex: present / absent R/ L (optional task)
Table 2:
References:
Albert, S. M., Murphy, P. L., Del Bene, M., Rowland, L. P., & Mitsumoto, H. (2001). Incidence and predictors of PEG placement in ALS/MND. Journal of the Neurological Sciences, 191(1–2), 115–119. doi:10.1016/S0022-510X(01)00614-1
D’Ottaviano, F. G., Linhares Filho, T. A., Andrade, H. M. T. de, Alves, P. C. L., & Rocha, M. S. G. (2013). Vídeoendoscopia da deglutição na esclerose lateral amiotrófica. Brazilian Journal of Otorhinolaryngology, 79(3), 349–353. doi:10.5935/1808-8694.20130061
Katzberg, H. D., & Benatar, M. (2011). Enteral tube feeding for amyotrophic lateral sclerosis/motor neuron disease. The Cochrane database of systematic reviews, (1), CD004030. doi:10.1002/14651858.CD004030.pub3
Kühnlein, P., Gdynia, H.-J., Sperfeld, A.-D., Lindner-Pfleghar, B., Ludolph, A. C., Prosiegel, M., & Riecker, A. (2008). Diagnosis and treatment of bulbar symptoms in amyotrophic lateral sclerosis. Nature clinical practice. Neurology, 4(7), 366–374. doi:10.1038/ncpneuro0853
Langmore, S. E., Kasarskis, E. J., Manca, M. L., & Olney, R. K. (2006). Enteral tube feeding for amyotrophic lateral sclerosis/motor neuron disease. The Cochrane database of systematic reviews, (4), CD004030. doi:10.1002/14651858.CD004030.pub2
Radunović, A., Mitsumoto, H., & Leigh, P. N. (2007). Clinical care of patients with amyotrophic lateral sclerosis.The Lancet Neurology, 6(10), 913–925. doi:10.1016/S1474-4422(07)70244-2
Robarge, K. Communication management in Amyotrophic Lateral Sclerosis: The role of the speech-language pathologist during disease progression. The Internet Journal of Allied Health Science and Pratice. April 2009, Volume 7 Number 2.
Albert, S. M., Murphy, P. L., Del Bene, M., Rowland, L. P., & Mitsumoto, H. (2001). Incidence and predictors of PEG placement in ALS/MND. Journal of the Neurological Sciences, 191(1–2), 115–119. doi:10.1016/S0022-510X(01)00614-1
D’Ottaviano, F. G., Linhares Filho, T. A., Andrade, H. M. T. de, Alves, P. C. L., & Rocha, M. S. G. (2013). Vídeoendoscopia da deglutição na esclerose lateral amiotrófica. Brazilian Journal of Otorhinolaryngology, 79(3), 349–353. doi:10.5935/1808-8694.20130061
Katzberg, H. D., & Benatar, M. (2011). Enteral tube feeding for amyotrophic lateral sclerosis/motor neuron disease. The Cochrane database of systematic reviews, (1), CD004030. doi:10.1002/14651858.CD004030.pub3
Kühnlein, P., Gdynia, H.-J., Sperfeld, A.-D., Lindner-Pfleghar, B., Ludolph, A. C., Prosiegel, M., & Riecker, A. (2008). Diagnosis and treatment of bulbar symptoms in amyotrophic lateral sclerosis. Nature clinical practice. Neurology, 4(7), 366–374. doi:10.1038/ncpneuro0853
Langmore, S. E., Kasarskis, E. J., Manca, M. L., & Olney, R. K. (2006). Enteral tube feeding for amyotrophic lateral sclerosis/motor neuron disease. The Cochrane database of systematic reviews, (4), CD004030. doi:10.1002/14651858.CD004030.pub2
Radunović, A., Mitsumoto, H., & Leigh, P. N. (2007). Clinical care of patients with amyotrophic lateral sclerosis.The Lancet Neurology, 6(10), 913–925. doi:10.1016/S1474-4422(07)70244-2
Robarge, K. Communication management in Amyotrophic Lateral Sclerosis: The role of the speech-language pathologist during disease progression. The Internet Journal of Allied Health Science and Pratice. April 2009, Volume 7 Number 2.